 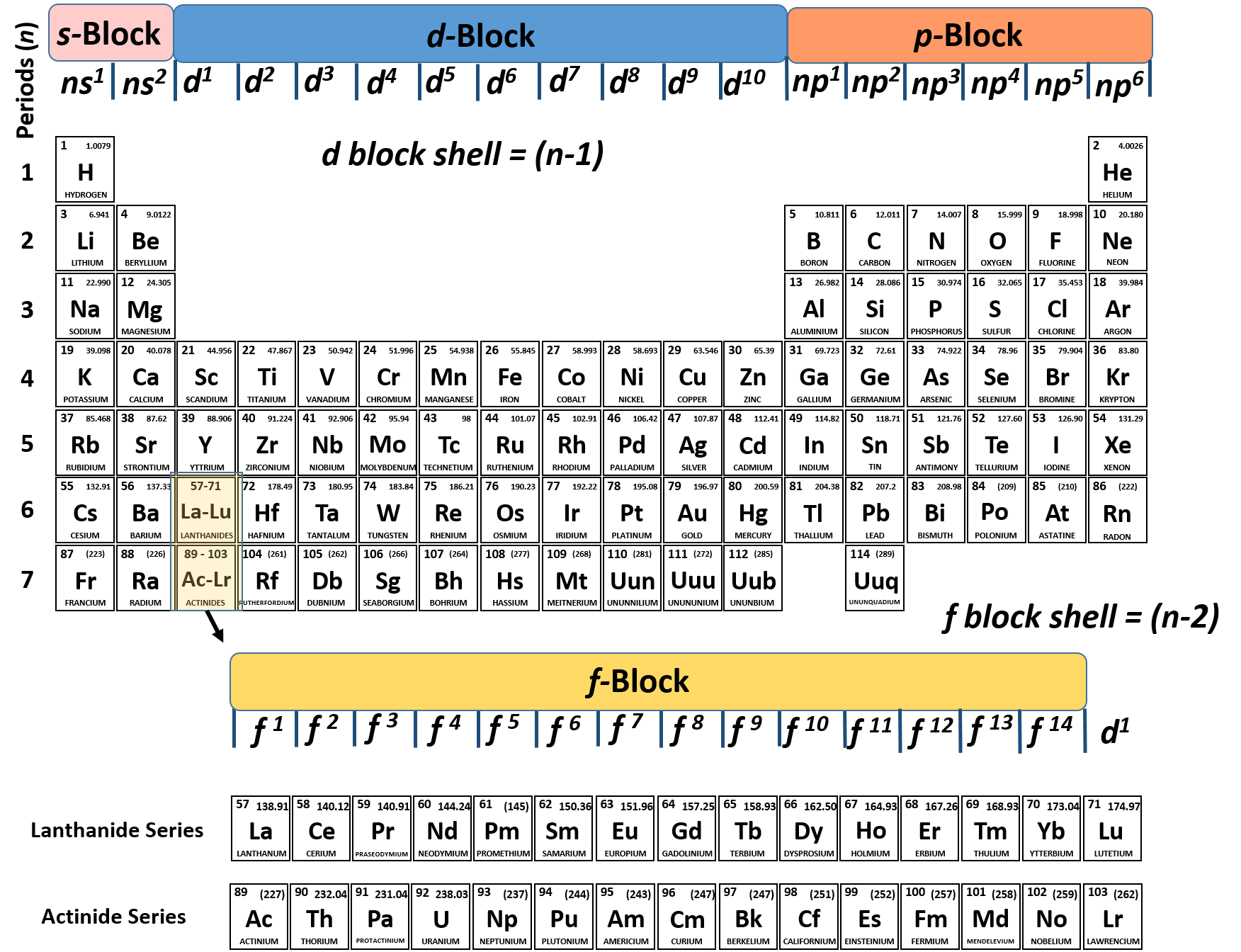
These elements’ electron configurations always end with one or more “f electrons. The outermost orbital shell of an atom is called its valence shell, and the electrons in the valence shell are valence electrons. The “f block” elements are those at the bottom of the periodic table that we call the lanthanide and actinide groups. 
For example, the all of the elements in group 3 beginning with scandium, all end in “d 1”. The transition elements are called “d block” elements and always end with 1 or more “d electrons”. All of the elements in group 16, beginning with oxygen, end with “p 4”. “P block” elements are all those in groups 13-18 and always end with 1 or more “p” electrons.įor example, all the elements in group 13, beginning with boron, end with “p 1”. For brevity, many chemists record the electron configuration of an atom by giving only its outermost subshell, like 4 s 1 for potassium or 4 s 2 for calcium. These 2 groups are know as “s block” elements. The alkali metal electron configurations (group 1) always end with “s 1” and the alkaline earth metals (group 2) always end with “s 2”. The periodic table can be broken into “blocks” that show what the last electron added to the electron configuration is. It will be helpful now to look at electron configurations also in the context of the periodic table. For the undiscovered eighth-row elements, mixing of configurations is expected to be very important, and sometimes the result can no longer be well-described by a single configuration.Electron Configurations and the Periodic TableĮlectron configurations and the periodic table In many cases, multiple configurations are within a small range of energies and the irregularities shown below do not necessarily have a clear relation to chemical behaviour. For representative elements, the number of valence electrons equals. Note that these electron configurations are given for neutral atoms in the gas phase, which are not the same as the electron configurations for the same atoms in chemical environments. For example, hydrogen has only one electron-its valence electron. However there are numerous exceptions for example the lightest exception is chromium, which would be predicted to have the configuration 1s 2 2s 2 2p 6 3s 2 3p 6 3d 4 4s 2, written as 3d 4 4s 2, but whose actual configuration given in the table below is 3d 5 4s 1. The valence electrons (here 3s 2 3p 3) are written explicitly for all atoms.Įlectron configurations of elements beyond hassium (element 108) have never been measured predictions are used below.Īs an approximate rule, electron configurations are given by the Aufbau principle and the Madelung rule. Here refers to the core electrons which are the same as for the element neon (Ne), the last noble gas before phosphorus in the periodic table. For phosphorus (element 15) as an example, the concise form is 3s 2 3p 3. For each atom the subshells are given first in concise form, then with all subshells written out, followed by the number of electrons per shell. Write the electron structure of the +1 cation of thallium. 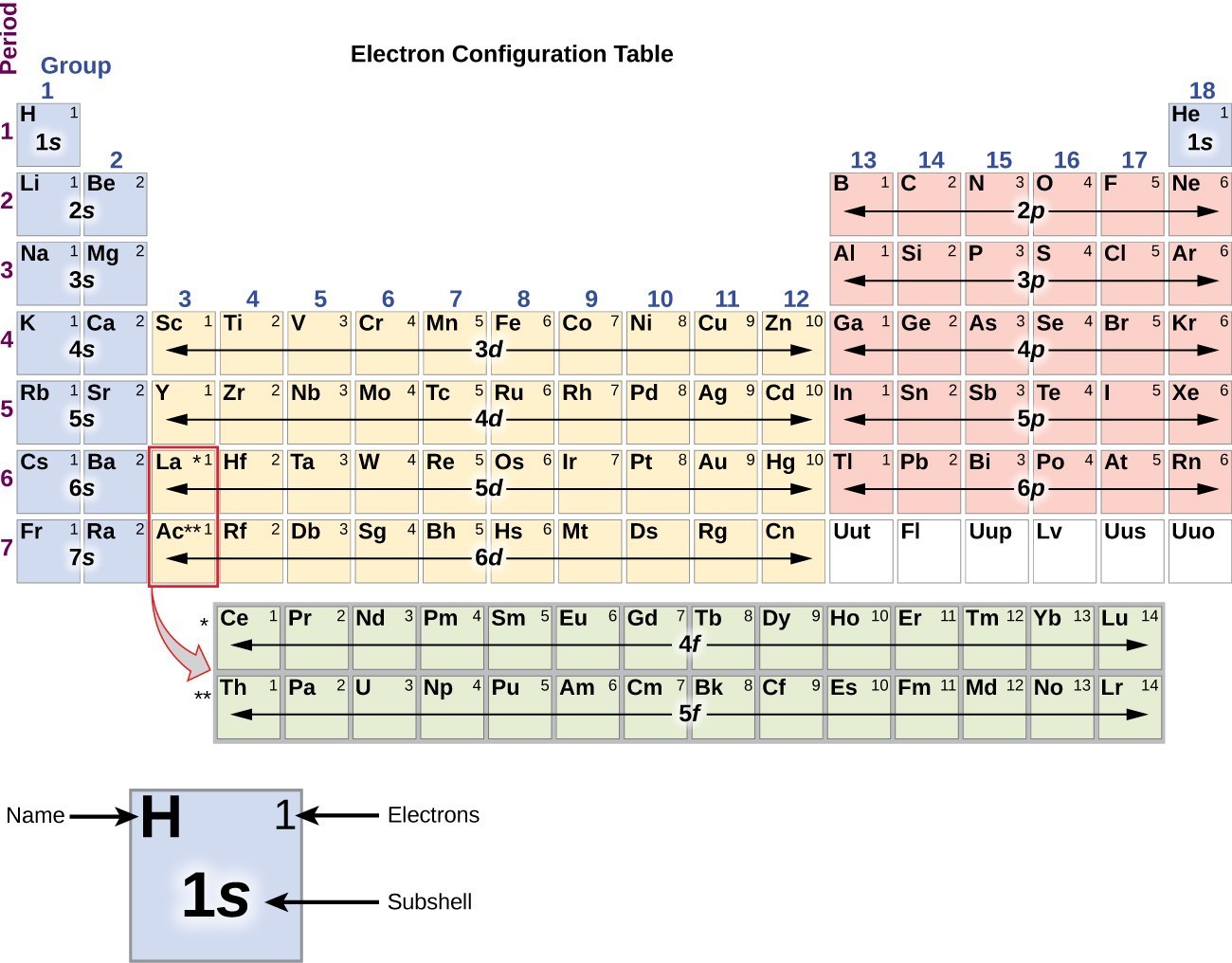
Thallium has two possible cationic forms, +1 and +3. This page shows the electron configurations of the neutral gaseous atoms in their ground states. Thallium was used as a poison in the Agatha Christie mystery story The Pale Horse. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
